Bone Remodeling: The Biological Cycle That Decides Implant Fate
Leggi in Italiano
In brief — Bone remodeling is the continuous process through which bones regenerate, repair microscopic damage, and adapt to mechanical stress. Three coordinated cell types orchestrate this cycle: osteoclasts demolish old bone, osteoblasts deposit new tissue, and osteocytes supervise the process. In implantology, this remodeling determines osseointegration and implant success.
In breve (IT) — Il rimodellamento osseo è il processo continuo attraverso cui le ossa si rigenerano, riparano danni microscopici e si adattano alle sollecitazioni. Tre cellule coordinano questo ciclo: osteoclasti demoliscono l’osso vecchio, osteoblasti ne depositano di nuovo, osteociti supervisionano tutto. Per l’implantologia, questo rimodellamento determina l’osteointegrazione e il successo dell’impianto.
Your bones are rebuilding themselves right now, as you read these lines.
They are not static structures. They are living tissue in constant renewal. This holds true everywhere in the skeleton, from alveolar bone to long bones. The process is called bone remodeling.
Perpetual Maintenance
Think of your bones as a building that requires continuous maintenance.
Damaged sections are removed and replaced with new material, without compromising the load-bearing structure. This happens in your skeleton every day, every hour.
Bone remodeling involves three cell types working in perfect synchrony. Osteoclasts are the demolition crew: they remove old or damaged bone.
Osteoblasts are the builders: they deposit new bone tissue.
Osteocytes are the supervisors: they coordinate the entire process and maintain balance. These cells form a sophisticated communication network that allows bone to respond to mechanical and metabolic stimuli.
The Remodeling Cycle
The process follows a precise sequence that begins with activation.
Osteocytes, embedded within the bone matrix yet always biologically dynamic, sense damage or mechanical stress in the surrounding tissue.
When they detect microfractures or aged tissue, they release signaling molecules such as ATP and RANKL. It is a refined biological alarm system.
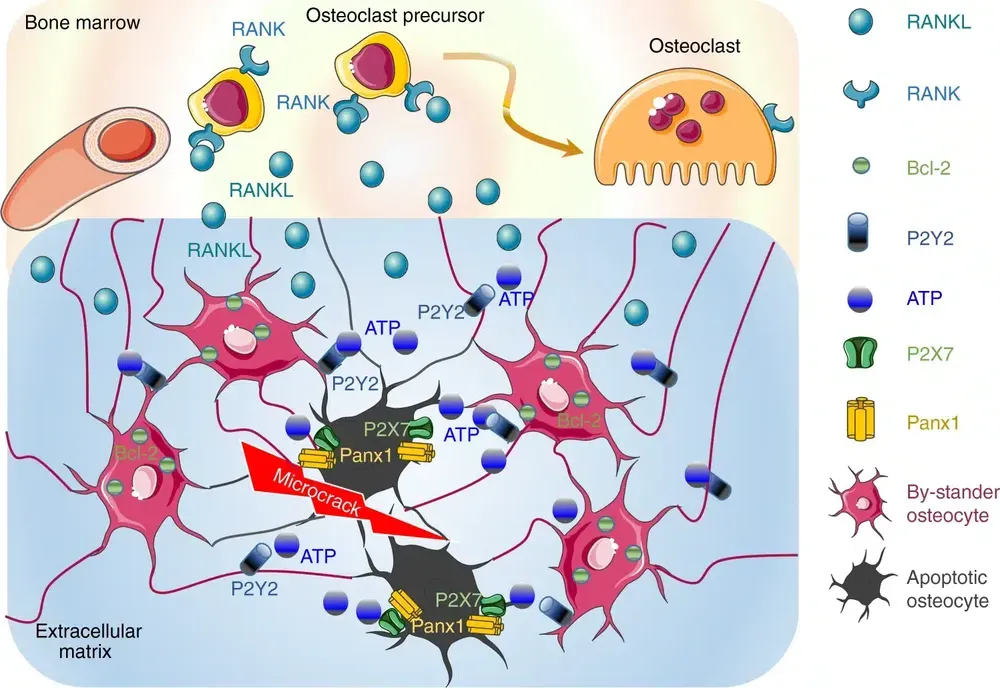
Next comes resorption.
Osteoclasts arrive and begin digesting damaged bone, creating small cavities called Howship’s lacunae. This phase lasts approximately two to four weeks.
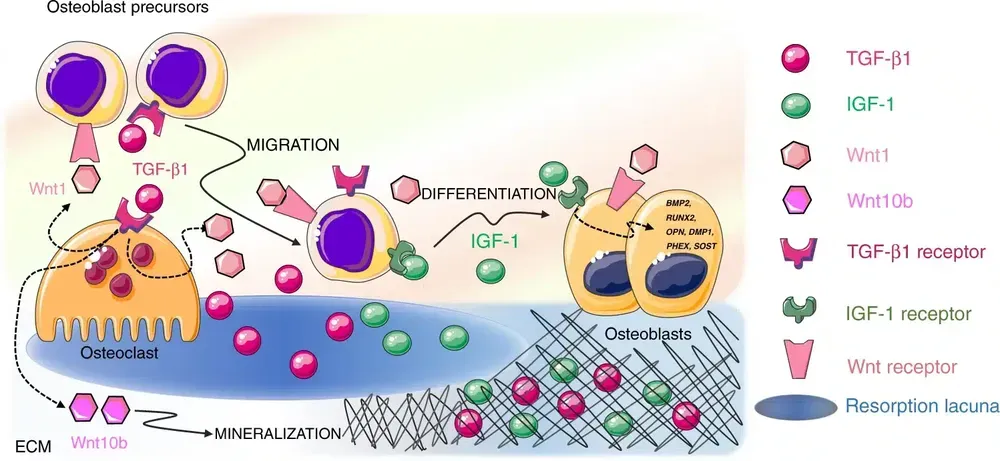
Then comes reversal: specialized cells called reversal cells prepare the surface for the next phase, cleaning debris and creating optimal conditions for new bone formation.
In the immediately following formation phase, osteoblasts deposit new bone tissue (osteoid) that promptly mineralizes.
This phase can last three to four months.
Finally, quiescence: some osteoblasts become osteocytes, remaining embedded within the new bone, while others become the lining cells that cover the bone surface (periosteal cells).
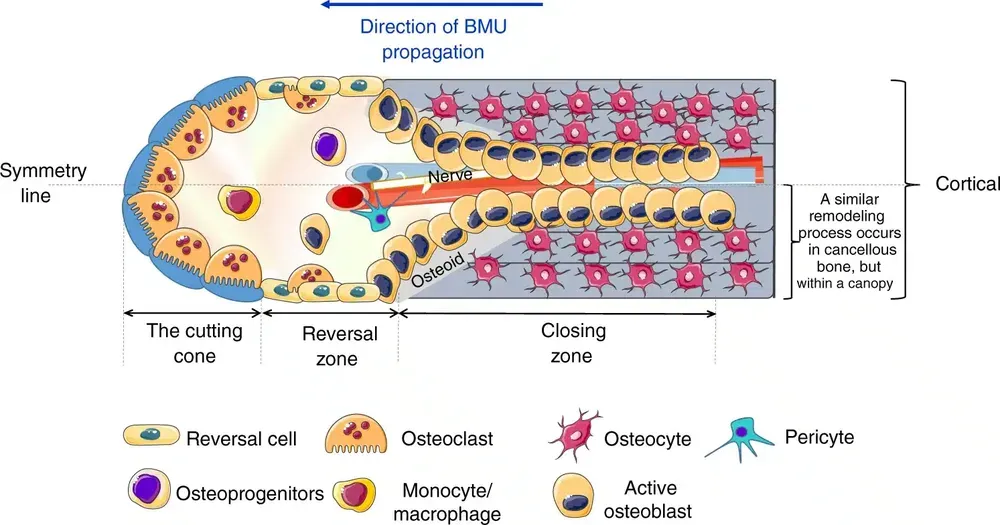
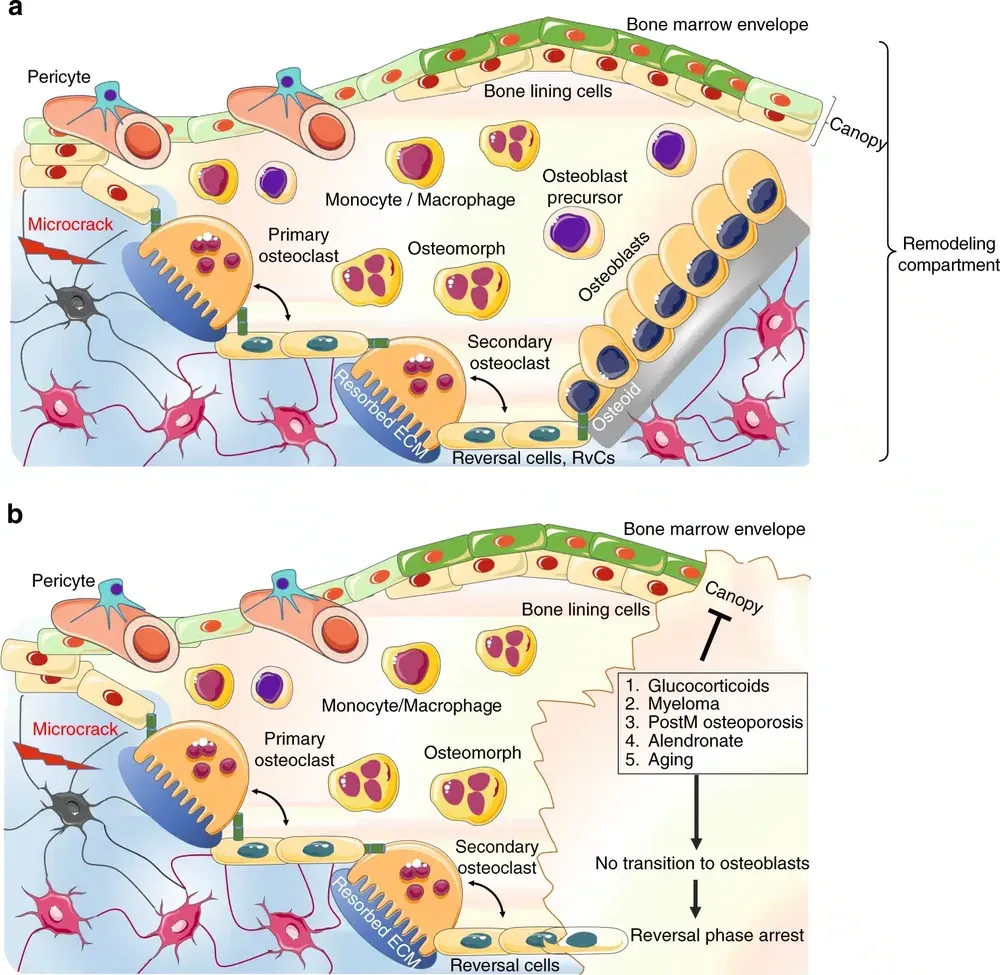
BMU: The Operating Unit
Bone cells do not work randomly.
They form a functional unit called the BMU (Basic Multicellular Unit). It is a team of specialized workers moving through bone, renewing it section by section.
A fascinating feature is the creation of the Bone Remodeling Compartment (BRC): a protected space where all the work takes place, separated from the rest of the bone marrow by a structure called the canopy.
It is like a fenced construction site inside an inhabited building.
Why It Matters
Bone remodeling serves to repair microdamage from daily use, to adapt bone structure to mechanical loads, to maintain calcium homeostasis in blood, and to renew aged bone tissue.
But for those of us in implantology, there is another crucial function: osseointegration of dental implants.
When an implant is placed in bone, remodeling determines its fate. Primary stability depends on mechanics. Secondary stability depends on biology. And biology is governed by remodeling.
When Balance Breaks Down
Aging causes resorption to outpace formation. Menopause, with declining estrogen levels, accelerates bone loss. Osteoporosis disrupts the system’s equilibrium. Certain drugs, particularly bisphosphonates, profoundly alter remodeling dynamics: they block osteoclasts, reducing resorption but also the bone’s capacity to renew and adapt.
For a deeper look at how these factors affect implantology, I have written a dedicated article on biological mechanisms of implant failure.
Remodeling and Implantology
Understanding bone remodeling has profound implications for implant practice. When we place an implant, we are not simply fixing a screw into bone. We are initiating a complex biological dialogue with the patient’s organism.
The screw concept is a losing one. We must move beyond the cliché of a screw in bone, because bone is not a plank of wood, and an implant that relies solely on its threads will lose stability within days and become encapsulated in fibrous tissue like any foreign body (see below).The implant surface is recognized as “foreign” by the immune system. Macrophages arrive first and their polarization toward the M2 phenotype (pro-regenerative) rather than M1 (pro-inflammatory) determines whether bone will embrace the implant or isolate it with fibrous tissue.
If myofibroblasts from fibrous tissue prevail, the implant will fail.
Bone quality influences remodeling. Dense bone (type A1 in the new Wang classification) will react differently than trabecular bone (type C3).
Vitamin D3 modulates the process: insufficient levels compromise mineralization of new bone.
The Bonebenders Method
The bone expansion techniques I have used for over 30 years are founded on understanding remodeling.
Instead of adding heterologous bone from outside and waiting for it to remodel, we stimulate native bone to expand and regenerate through its own natural mechanisms.A meta-analysis of 1,400 implants demonstrated a 98.1% success rate with this technique. It is not magic. It is biology respected.
When we expand a thin bone ridge, we create a controlled mechanical stimulus. Osteocytes sense it and activate remodeling.
Osteoblasts deposit new bone in tension zones. The result is vital, vascularized bone capable of responding to the functional stimuli of mastication.
Therapeutic Prospects
Increasingly detailed understanding of remodeling is opening new paths. There are drugs that specifically modulate osteoclast activity, such as denosumab, and therapies that stimulate osteoblasts.
The most promising approaches exploit cell communication through extracellular vesicles.
For implantology, the frontier is modulation of the peri-implant microenvironment: surfaces that guide macrophage polarization, treatments that optimize the osteoblastic response, protocols that respect the biological timing of remodeling.
Conclusion
Bone remodeling is an extraordinary example of how the body maintains and renews its tissues.
Approximately 10% of the skeleton is renewed every year.
In roughly ten years, we have an entirely new skeleton.
For those who place implants, this knowledge is not academic. It is the difference between working with biology or against it. Bones are not mere support structures. They are living tissues that respond, adapt, heal. To respect remodeling is to respect life.
This post is based on the scientific article “Bone remodeling: an operational process ensuring survival and bone mechanical competence” by Bolamperti et al., published in Bone Research (2022).
Related Articles
To explore the topics covered in this article: The osteocyte connectome, Macrophages in osseointegration, The new alveolar bone classification, The truth about primary stability.
FAQ
What is bone remodeling and why does it matter?
Bone remodeling is the continuous process through which bones regenerate and adapt to mechanical loads, repairing microdamage and maintaining calcium homeostasis. It is essential for skeletal health and, in implantology, determines whether a dental implant succeeds or fails.
How do the cells involved in bone remodeling work?
The main cells are osteoclasts, which demolish old bone; osteoblasts, which deposit new bone tissue; and osteocytes, which supervise the entire process by coordinating activity and maintaining balance.
What are the phases of the bone remodeling cycle?
The cycle includes: activation, where osteocytes signal damage or mechanical stress; resorption, where osteoclasts digest damaged bone; reversal, where cells prepare the surface for new bone; formation, where osteoblasts deposit new tissue; and quiescence, when some cells become osteocytes or lining cells.
Why is bone remodeling crucial in implantology?
Remodeling determines the secondary stability of dental implants. It dictates whether an implant will integrate with bone or be isolated by fibrous tissue. The biological response of bone to the implant depends on the correct functioning of this process.
What are the future therapeutic prospects for bone remodeling?
Future therapies focus on specific modulation of the bone microenvironment, using drugs such as denosumab and innovative technologies like extracellular vesicles to improve cell communication and promote bone regeneration, including in implantology.
FAQ
- What is bone remodeling and why does it matter?
- Bone remodeling is the continuous process through which bones regenerate and adapt to mechanical loads, repairing microdamage and maintaining calcium homeostasis. It is essential for skeletal health and, in implantology, determines whether a dental implant succeeds or fails.
- How do the cells involved in bone remodeling work?
- The main cells are osteoclasts, which demolish old bone; osteoblasts, which deposit new bone tissue; and osteocytes, which supervise the entire process by coordinating activity and maintaining balance.
- What are the phases of the bone remodeling cycle?
- The cycle includes: activation, where osteocytes signal damage or mechanical stress; resorption, where osteoclasts digest damaged bone; reversal, where cells prepare the surface for new bone; formation, where osteoblasts deposit new tissue; and quiescence, when some cells become osteocytes or lining cells.
- Why is bone remodeling crucial in implantology?
- Remodeling determines the secondary stability of dental implants. It dictates whether an implant will integrate with bone or be isolated by fibrous tissue. The biological response of bone to the implant depends on the correct functioning of this process.
- What are the future therapeutic prospects for bone remodeling?
- Future therapies focus on specific modulation of the bone microenvironment, using drugs such as denosumab and innovative technologies like extracellular vesicles to improve cell communication and promote bone regeneration, including in implantology.
References
Looking for a specialist?
Innesto Osseo a Frosinone →Tipi di innesto, tecniche e indicazioni per ricostruire l'osso perduto
Need a professional opinion?
Book an appointment at Dr. Bruschi's practice in Frosinone. First visit includes full diagnosis and personalised treatment plan.
Stai valutando un impianto dentale?
Ho scritto una guida in 8 capitoli che spiega tutto quello che un paziente dovrebbe sapere prima di sedersi in poltrona. Niente marketing — solo fatti, casi studio e una checklist per fare le domande giuste.
Scarica la guidaStay Updated
New articles on periodontology, implantology and oral surgery — delivered to your inbox.
Comments
Loading comments...
Leave a comment